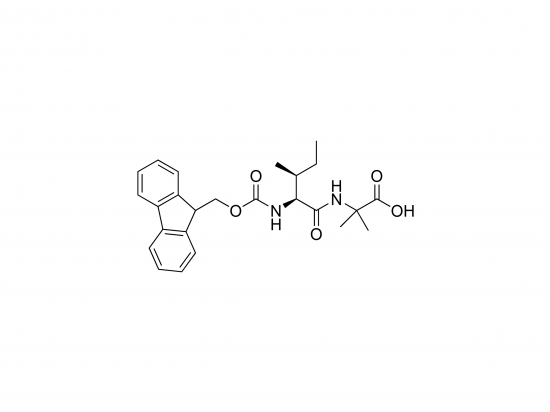
Fmoc-Ile-Aib-OH is a dipeptide building block widely used in peptide synthesis to induce specific secondary structures such as α-helices and 3₁₀-helices. It couples L-isoleucine (Ile) with α-aminoisobutyric acid (Aib). The N-terminus is protected with the 9-fluorenylmethyloxycarbonyl (Fmoc) group, enabling efficient solid-phase peptide synthesis (SPPS). Aib, an unnatural and sterically hindered amino acid, promotes helical conformations by imposing conformational constraints, making this building block valuable for designing peptides with defined structures in drug discovery, biomaterials, and nanotechnology. It is generally supplied as a white solid with high purity.
Appearance
-
White to off-white crystalline solid or powder
-
Odorless
-
Non-hygroscopic or slightly hygroscopic
Source
-
Synthesized by standard peptide chemistry methods
-
Commercially available from suppliers such as Sigma-Aldrich, Bachem, Novabiochem, Iris Biotech
-
Typically produced by coupling Fmoc-Ile-OH to Aib-OH using coupling reagents (DIC, HATU), followed by purification
Molecular Weight and Structure
-
Molecular formula: C₂₂H₃₂N₂O₅
-
Molecular weight: 404.51 g/mol
-
IUPAC name: (2S)-2-[[(9H-fluoren-9-yl)methoxy]carbonylamino]-3-methylpentanoyl-2-methylalanine
-
Structure: Fmoc-Ile-Aib-OH
-
SMILES (partial): CCC@HC@HC(=O)C(C)(C)N (simplified)
Biological Activity
-
No inherent biological activity; serves as a peptide synthesis building block
-
Enhances α-helical structure and stability when incorporated in peptides
-
May improve cell permeability and enzymatic resistance of peptides
Purity and Microbial Contamination
-
Purity: >98% by HPLC
-
Enantiomeric purity: >99% for L-Isoleucine
-
Water content: <1% by Karl Fischer titration
-
Microbial contamination: Controlled to <100 CFU/g or per mg peptide
-
Endotoxin levels: <10 EU/mg peptide
Identity and Quality Control
| Test | Method | Acceptance Criteria |
|---|---|---|
| Mass spectrometry | ESI-MS | [M+H]+ within ±1 Da of expected mass |
| HPLC | RP-HPLC, C18 | Single peak with >98% area |
| NMR | ¹H and ¹³C NMR | Spectrum consistent with structure |
| Optical Rotation | Polarimetry | Specific rotation within limits |
| Melting Point | Capillary method | Within narrow range |
Shelf Life and Storage
-
Store at 2–8 °C or –20 °C
-
Protect from light and moisture
-
Typical shelf life: 2 years from manufacture if stored correctly
-
Avoid repeated freeze-thaw cycles
Applications
-
Peptide synthesis as a helix-inducing building block
-
Drug design and development
-
Biomaterials and tissue engineering
-
Nanotechnology and self-assembling peptides
-
Studies of peptide folding and stability
Key Characteristics
-
Fmoc-protected for use in SPPS
-
Contains L-isoleucine and Aib amino acids
-
Aib induces helical conformations, enhancing peptide stability
-
Improves cell permeability and resistance to degradation
-
Well-defined chemical structure and high purity
Citation
-
Toniolo C, Benedetti E. Structures of polypeptides containing alpha,alpha-disubstituted alpha-amino acids. Acc Chem Res. 1991;24(12):400-6. https://doi.org/10.1021/ar00012a004
-
Karle IL, Balaram P. Structural characteristics of alpha/alpha-disubstituted amino acid residues in peptides. Biochemistry. 1990;29(29):6747-56. https://pubmed.ncbi.nlm.nih.gov/2202542/
-
Venkatachalam CM. Stereochemical criteria for polypeptides and proteins. V. Conformation of a system of three linked peptide units. Biopolymers. 1968;6(10):1425-44. https://pubmed.ncbi.nlm.nih.gov/5678563/
-
DeGrado WF, Lear JD, Eisenberg D, Taylor WR. New proteins from the test tube. Science. 1989;243(4899):622-8. https://pubmed.ncbi.nlm.nih.gov/2643078/
-
Woolfson DN, Bartlett GJ. De novo design of self-assembling peptides. Chem Commun. 2008;(45):5773-97. https://doi.org/10.1039/B806998H
-
Gellman SH. Foldamers: a manifesto. Acc Chem Res. 1998;31(4):173-80. https://doi.org/10.1021/ar960299r
-
Nowick JS. Modular design of self-assembling peptide derivatives. Acc Chem Res. 1999;32(4):287-96. https://doi.org/10.1021/ar970
Reviews
There are no reviews yet.